Any content you receive is for information purposes only. Always conduct your own research.*Sponsored
See Why Market Crux Just Put (NDRA) On This Morning's Watchlist—Thursday, February 26, 2026
Don't Miss The Next Breakout—Get Real-Time Alerts Sent Directly
To Your Phone. Up To 10X Faster Than Email.
Take A Look AT (NDRA) While It's Still Early…
February 26, 2026
It's Almost Time | (NDRA) is Topping Our Watchlist Right Now—Here's Why Dear Reader, A quiet crisis is spreading in plain sight—and most people won't realize they're affected until serious damage is already done. What makes it more alarming is that the warning signs often stay hidden during routine visits, even as the problem accelerates worldwide. Yet one emerging breakthrough is aiming to change that by bringing a level of insight that has historically been reserved for the most expensive hospital equipment. If it works as intended, it could turn a complicated, hard-to-access measurement into something that feels as routine as a basic check-up. ENDRA Life Sciences Inc. (NASDAQ: NDRA) is stepping into this diagnostic void with a pioneering technology that aims to make high-end imaging as accessible as a standard check-up. And this is just one of the reasons why (NDRA) is topping our watchlist this morning—Thursday, February 26, 2026. But keep in mind, (NDRA) has less than 1.2M shares listed as available for the public. When companies have small floats like this, the potential exists for big moves if demand begins to shift. And it's not just the technology turning heads—because a fresh analyst view is putting a striking spotlight on (NDRA) right now. Analyst Target Suggests 750% Upside Potential From Recent Range
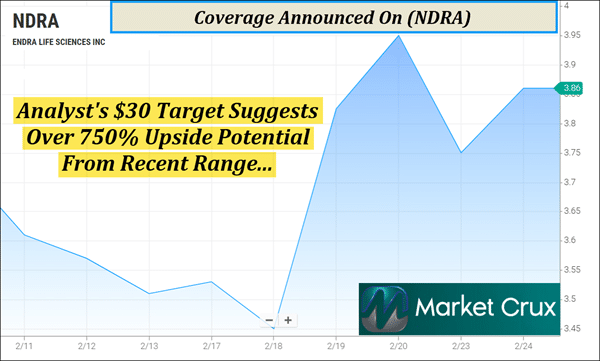
The disconnect between the company's market capitalization and its technological potential has been noted by institutional researchers. Edward Woo, CFA of Ascendiant Capital Markets, recently set a $30 target on (NDRA). Given its recent range around $3.50, this target suggests a potential upside of over 750%. This valuation is based on the massive unmet need in the liver disease space and the ability of TAEUS® to serve as a high-margin "razor and blade" business model, where the sale of the hardware leads to recurring service and software revenue. Company Overview: Pioneering the Future of Thermo-Acoustic Imaging
ENDRA Life Sciences Inc. (NASDAQ: NDRA) is the developer of the proprietary Thermo Acoustic Enhanced UltraSound (TAEUS®) technology. This innovative platform is being engineered to visualize tissue in ways that were previously the exclusive domain of Magnetic Resonance Imaging (MRI), but at the point of patient care. While traditional ultrasound is highly effective at displaying physical structures, it has historically lacked the sensitivity required to accurately quantify tissue fat—a critical metric for diagnosing and managing liver disease. The company's primary focus is the measurement of fat in the liver to assess and monitor Steatotic Liver Disease (SLD) and Metabolic Dysfunction-Associated Steatohepatitis (MASH). These conditions are characterized by the accumulation of excess fat in the liver, which, if left unmonitored, can lead to inflammation, fibrosis, and eventual liver failure. Currently, the "gold standard" for fat quantification is MRI-PDFF, an expensive and time-consuming procedure that is often inaccessible for routine monitoring. ENDRA Life Sciences Inc. (NASDAQ: NDRA) intends to solve this by offering a device that is roughly 1/50th the cost of an MRI and can be integrated into the workflow of a standard physician's office. Beyond the liver, the TAEUS® platform has been designed as a versatile "imaging engine." The company is exploring additional clinical applications, including the monitoring of tissue temperature during energy-based surgical procedures. This flexibility, backed by a global portfolio of over 80 issued patents, provides a significant competitive moat. As healthcare systems worldwide move toward decentralized care and non-invasive diagnostic models, (NDRA) is striving to become the new standard for metabolic disease detection. The $66B Global Liver Disease Diagnostics Market

The scale of ENDRA Life Sciences Inc. (NASDAQ: NDRA) is underscored by the rapid expansion of the liver disease diagnostics market. Projections indicate this sector will grow by over 60%, climbing from approximately $40B in 2025 to exceed $66B by 2033. This growth is primarily driven by a global emphasis on non-invasive testing methods and the rising prevalence of liver-related disorders such as fibrosis, cirrhosis, and hepatitis. Key market contributors include intensified efforts by leading companies to develop sensitive, rapid testing methods tailored for diverse demographics. Furthermore, shifts in lifestyle factors—including increased alcohol consumption and poor dietary habits—are projected to influence the market positively as the need for early detection becomes more acute. Within this environment, (NDRA)'s TAEUS® device is emerging as a critical tool for healthcare providers who need to identify patients at risk before they progress to irreversible stages of disease. Clinical Validation: Matching the Gold Standard
For any new diagnostic technology, clinical validation is the ultimate hurdle. In late 2025, ENDRA Life Sciences Inc. (NASDAQ: NDRA) achieved a major milestone by releasing feasibility study data that showed its TAEUS® system matches the performance of MRI-PDFF at key clinical thresholds. Specifically, at liver fat cutoffs of 12-22%, the device demonstrated high accuracy, identifying the transition from mild to moderate and severe disease. This performance is particularly significant for MASLD/MASH clinical trials. Pharmaceutical companies developing treatments for these conditions currently rely on expensive MRI scans to screen and monitor participants. A low-cost, point-of-care alternative like TAEUS® could significantly reduce the costs of drug development and enable more frequent longitudinal monitoring. A second feasibility study is already underway in Canada, further strengthening the clinical dossier as the company prepares for FDA engagement in 2026. Strategic Financial Initiatives and Balance Sheet Strength
To support its commercialization efforts, ENDRA Life Sciences Inc. (NASDAQ: NDRA) has recently undertaken several significant fiscal initiatives. Additionally, the company launched a "Next-Generation Treasury Strategy," deploying a portion of its capital into an institutional-grade reward-generating program designed to create non-dilutive financial returns. Operational discipline is also a core focus. The company reported a 30% decrease in ca-sh burn from operations in Q3 2025, reaching $1.2M compared to $1.7M in the prior year. This leaner operating model, combined with the new capital influx, provides a runway to complete critical clinical studies and pursue De Novo filing with the FDA. With zero long-term debt as of the end of 2025, NDRA is prioritizing financial flexibility as it transitions from R&D to market entry. Management and Technical Expertise
The leadership team at ENDRA Life Sciences Inc. (NASDAQ: NDRA) brings a wealth of experience in medical device development and regulatory affairs. CEO Alex Tokman, who took the helm in late 2024, previously spent over a decade at GE Healthcare, where he drove innovation in diagnostic imaging. The board also includes veterans like Michael Harsh, a former Chief Technology Officer at GE Healthcare with 36 years of experience in diagnostic imaging. This deep industry knowledge is vital for navigating the complex regulatory and commercial hurdles inherent in launching a disruptive medical technology. 7 Reasons Why (NDRA) is Topping Our Watchlist This Morning—Thursday, February 26, 2026…
1. Small Float: With fewer than 1.2M shares listed as available for the public, (NDRA)'s small float could have the potential to see big moves if demand begins to shift. 2. Analyst Target: Ascendiant's $30 target suggests over 750% upside potential for (NDRA) from its recent $3.50 range. 3. MRI-Level Data: The proprietary TAEUS® platform developed by (NDRA) is engineered to match MRI-PDFF performance at key liver fat thresholds in feasibility studies. 4. Cost Advantage: By offering scans estimated at roughly 1/50th the cost of an MRI, (NDRA) is positioning its system for broader point-of-care adoption. 5. $66B Market: The liver disease diagnostics sector is projected to expand from about $40B in 2025 to over $66B by 2033, placing (NDRA) within a rapidly growing segment. 6. Patent Portfolio: Backed by more than 80 issued patents worldwide, (NDRA) has built a substantial intellectual property foundation around its thermo-acoustic imaging technology. 7. Lean Balance Sheet: With a reported 30% reduction in operational ca-sh burn year-over-year and zero long-term debt at the end of 2025, (NDRA) is advancing toward FDA engagement from a position of financial discipline. Take A Look AT (NDRA) While It's Still Early…

(NDRA) is one of those little-known companies where multiple pieces appear to be lining up at the same time. You have a small public float (under 1.2M shares), analyst coverage pointing to a $30 target that suggests over 750% upside potential from its recent $3.50 range, and a platform—TAEUS®—built to deliver MRI-PDFF–level liver fat measurement at key clinical thresholds based on feasibility data. Add in a scan cost profile estimated at roughly 1/50th of an MRI, a liver diagnostics market projected to reach $66B by 2033, and a defensible moat of 80+ issued patents, and it's easy to see why (NDRA) could start to stand out. On top of that, (NDRA) has been tightening operations with a reported 30% reduction in ca-sh burn and no long-term debt as of the end of 2025, while preparing for FDA engagement in 2026. We have all eyes on (NDRA) this morning—Thursday, February 26, 2026. Take a look at (NDRA) while it's still early. Also, keep a lookout for my next update, it could be hitting very soon. Sincerely, Gary Silver
Managing Editor,
Market Crux
| 




No comments:
Post a Comment
Keep a civil tongue.